Oral Rehydration Therapy: How it Works
Author: Roger M. Goodall
We are often asked to explain how ORT works - a question that can only be answered successfully by first considering some of the simple physiology of the normal intestine and then the changes that occur in a state of diarrhoeal disease. This is a basic discussion of the question written in reasonable non-technical terms to provide some of this interesting background information.
GLOSSARY OF SOME OF THE TERMS USED
METABOLITE
Simple components into which food is broken down by digestion and which are subsequently built up into complex materials of body tissues e.g. proteins which are broken down into their component amino-acids by digestion and then metabolized back into further proteins in the body.
ION
A single electrically charged particle into which the atoms or molecules of some substances dissociate when in solution, e.g. sodium chloride in the solid state consists of molecules containing one atom of sodium Na and one atom of chlorine Cl bound together NaCl - in solution in water the molecule splits into two ions (Na+) and (Cl-) each of which tends to be loosely bound to three or four molecules of water e.g. (H8O4Na) + and (H6O3Cl) - although for practical purposes they can be thought of as single ions Na+ and Cl- Positively charged ions e.g. Na+ are called CATIONS and Negatively charged ions e.g. Cl- are called ANIONS. The substances which show this dissociation into electrically charged ions are called ELECTROLYTES.
SOLUTE
A dissolved substance e.g. sodium chloride (the solute) dissolved in water (the solvent) to give a solution.
MOLARITY
If two different substances are in a solution they are said to be equal in molarity (equimolar) if they have equal numbers of molecules per litre of solution. The mass or weight of each solute is then proportionate to their respective molecular weights.
HYPERNATRAEMIA
The presence of an excess amount of sodium Na+ in the blood plasma (i.e. over 140 mmol/l.) NORMONATRAEMIC - is the presence of a normal level of sodium and HYPONATRAEMIC - lower than normal sodium level in the plasma.
UNICEF/WHO O.R.S
- Sodium Chloride 3.5 grams
- Sodium Bicarbonate 2.5 grams
- Potassium Chloride 1.5 grams
- Glucose 20 grams
to be dissolved in one litre of clean drinking water REFERENCE: The management of diarrhoea and use of oral rehydration therapy a Joint WHO/UNICEF statement.
THE PHYSIOLOGICAL PROCESS
In the normal healthy intestine, there is a continuous exchange of water through the intestinal wall - up to 20 litres of water is secreted and very nearly as much is reabsorbed every 24 hours - this mechanism allows the absorption into the bloodstream of soluble metabolites from digested food. Typical values for the daily gains and losses of water in an average man in a temperate climate are:
Intake |
Volume
ml per day |
Output |
Volume
ml per day |
| Drink |
1300 |
Urine |
1500 |
| Food |
850 |
Expired Air |
400 |
| Metabolic Water |
350 |
Skin |
500 |
| |
|
Faeces |
100 |
Total |
2500 |
Total |
2500 |
In a state of diarrhoeal disease the balance is upset and much more water is secreted than is reabsorbed causing a net loss to the body which can be as high as several litres a day. In addition to water, sodium is also lost. The body's store of sodium (in the form of sodium ions Na+) is almost entirely in solution in body fluids and blood plasma, i.e., extra cellular. By contrast 98% of the body's total potassium (K+) is held within cells, i.e. intracellular. Approximate concentrations of the principal ions in plasma, interstitial, and intracellular fluids in an average man are:
| |
Plasma |
Interstitial
fluid |
Intracellular
fluid |
| Cations (mmol per litre |
|
|
|
| Sodium |
140 |
144 |
10 |
| Potassium |
4 |
4 |
155 |
| Calcium |
2.5 |
2 |
1 |
| Magnesium |
1 |
1 |
15 |
| Anions (mmol per litre) |
|
|
|
| Chloride |
102 |
114 |
5 |
| Bicarbonate |
27 |
30 |
10 |
| Phosphate |
1 |
1 |
50 |
| Sulphate |
0.5 |
0.5 |
10 |
| Protein |
2 |
0.1 |
8 |
| Organic Anions |
3 |
6 |
2 |
The concentration of Na+ in the extracellular fluid has to be held to within close limits (135-150 mmol/l) for the proper functioning of the body. Normally, this sodium concentration is normally precisely controlled by the renal function. However in a state of dehydration water is conserved by anuria and the sodium regulation cannot work effectively. Thus continued diarrhoea causes rapid depletion of water and sodium, which is to say, a state of dehydration. If more than 10% of the body's fluid is lost death occurs. The approximate distribution of body water in an average man is:
| Compartment |
Volume
litres |
% of total
body water |
| Total body water |
42 |
100 |
| Extracellular |
17 |
40 |
| Plasma |
3.2 |
7.6 |
| Interstitial |
12.8 |
30 |
| Transcellular |
1 |
2.4 |
| Intracellular |
25 |
60 |
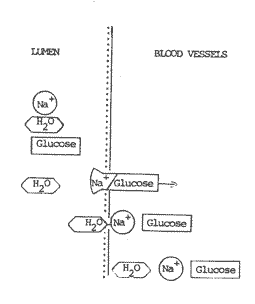
Simply giving a saline solution (water plus Na+) by mouth has no beneficial effect because the normal mechanism by which Na+ is absorbed by the healthy intestinal wall is impaired in the diarrhoeal state and if the Na+ is not absorbed neither can the water be absorbed. In fact, excess Na+ in the lumen of the intestine causes increased secretion of water and the diarrhoea worsens. If glucose (also called dextrose) is added to a saline solution a new mechanism comes into play. The glucose molecules are absorbed through the intestinal wall - unaffected by the diarrhoeal disease state - and in conjunction sodium is carried through by a co-transport coupling mechanism. This occurs in a 1:1 ratio, one molecule of glucose co-transporting one sodium ion (Na+). It was the discovery of this mechanism of co-transport of sodium and glucose which the Lancet described as "potentially the most important medical advance this century" ( ORT is in fact the practical realization of this potential). It should be noted that glucose does not co-transport water - rather it is the now increased relative concentration of Na+ across the intestinal wall which pulls water through after it. Several other molecules apart from glucose have a similar capacity to co-transport Na+ including:
 |
- aminoacids (e.g. glycine)
- dipeptides
- tripeptides
and the absorption of these molecules may occur independently of each other at different sites - thus their effect can be additive. Research is currently being carried on to utilize these additive effects to develop a multi-component "Super ORS".
|
Starch is metabolized in the intestine to glucose and therefore it has the same properties of enhancing sodium absorption, however it has an added advantage that it has less osmotic effect, which would act to pull water back into the lumen of the intestine.
THE COMPOSITION OF ORS.
In deciding the optimal composition of an oral rehydration solution the following considerations must apply:
- Sodium - losses of sodium in the stool range from 50-60 meg/l to well over 100 meg/l in cholera and in fact total body depletion of sodium may be higher than stool losses alone indicate. For this reason a Na+ concentration of 90 meg/l is considered an optimal figure for replenishing Na+ in dehydration from diarrhoea caused by any etiology and in all age groups from neonates to adults. For some years there was controversy over optimum concentration of sodium in oral rehydration fluids, which stemmed from the fact that in the early days of its use, particularly in USA, causes of hypernatraemia (excess sodium) occurred fairly frequently in infants given oral rehydration therapy. The apparently obvious answer was to assume that the sodium concentration in the oral rehydration fluid used was too high and to reduce it (even to as low as 25 or 30 meg/l). Unfortunately, the apparently obvious was not the correct answer - actually nearly all these children were being given high- solute infant formula which tended to make them hypernatraemic to start with and the oral rehydration solution used then contained excess glucose - up to 8% - which was added to provide extra nutritive calories. Unfortunately, the excess glucose caused osmotic diarrhoea which precipitated acute hypernatraemia in these children. The less obvious but correct answer was to reduce the glucose content - not the sodium. We now recognize that the sodium and glucose should be in a 1:1 ratio in terms of molarity. Experience has now shown that even hypernatraemic neonates with dehydration can be successfully rehydrated and made normonatraemic using the standard WHO / UNICEF ORS formula (with 90 meg/l Na+) when the water intake is sufficient to ensure normal kidney function and hence physiological regulation of the sodium concentration in the plasma.
Although ORS with a sodium content of around 50 meq/l is suffiient for maintenance of hydration of a normally will-nourished child with diarrhoea it would be inadequate for rehydration of a patient with a secretary diarrhoea (e.g., cholera) losing considerable sodium in the stool.
- Glucose should be close to equivalent with the Na+ content - it is 111 mmol/l in the WHO / UNICEF formula, which happens to be exactly 2%. It should be noted that if glucose is present in excess of 3% it will cause further losses of water through osmotic effects, this would also upset the electrolyte balance, since increased water losses will result in hypernatraemia.
- We have not yet given more than a passing mention to potassium. Although as we saw that 98% of the body's potassium is held within the cells, repeated diarrhoeal attacks over a period of time will cause a chronic loss of potassium. This results in muscular weakness, lethargy and anorexia. The typical distended abdomen of a chronically malnourished child is caused by loss of muscle tone in the abdominal wall largely due to chronic depletion of potassium. The kidneys are unable to conserve potassium as they do sodium, and there is a continuous obligatory loss of potassium of about 10 mmol daily in the urine, in addition to the larger losses in the stool. Potassium is not involved in any way in the sodium/glucose co-transport mechanism and is absorbed passively. Restoration of potassium levels is therefore achieved more slowly than sodium and water restoration. A potassium concentration of 20 mmol/l is considered optimal for the purpose. Simple mixtures of sugar , salt and water or starch, salt and water contain no potassium and cannot restore potassium depletion - hence these mixtures are an "incomplete" formula and further potassium supplementation is definitely necessary for a child who suffers repeated attacks of diarrhoea. A potassium-rich diet including, for example, bananas or coconut water can be helpful but an ORS solution containing potassium is therapeutically more effective. In order to produce a significant effect it is necessary to provide potassium-rich foods in reasonable large quantities over a period of time. Restoring a potassium deficit promotes a feeling of well-being and stimulates the appetite and activity of the child. If additional food is provided over several weeks an increase in weight gain will occur and the status of the child's health will improve markedly. Dietary intake is needed to achieve this.
- Electrolyte imbalance and fluid loss also causes metabolic acidosis. These effects are more critical in the case of infants, as their renal function is not fully developed and they have a large surface area in ratio to body weight and a higher metabolic rate. Acidosis is corrected by the addition of bicarbonate (or another base such as citrate) to the ORS formula.
Electrolyte content of stool in acute diarrhoea and the electrolyte and glucose content of ORS solution:
| |
Na+ |
K+ |
Cl- |
HCO3 |
| Cholera |
|
|
|
|
| adults |
140 |
13 |
104 |
44 |
| children (less than 5 yrs.) |
101 |
27 |
92 |
32 |
| Enteritis |
|
|
|
|
| children (less than 5 yrs.) |
56 |
25 |
55 |
14 |
| |
Glucose |
Na+ |
K+ |
cl- |
HCO3 |
| ORS Solution |
111 |
90 |
20 |
80 |
30 |
(values expressed as mmol/l)
The causative pathogens of diarrhoeal disease (which are very numerous, more than 30) in some cases not only produce the secretion of water and sodium but also damage the intestinal wall. The normal healthy intestine is covered on its inner surface with very numerous tiny hairs, or villi, the surface cells of which are involved in the absorption of metabolites from ingested food. There is a difference between the cells of the tips of the villi and the cells of the base in their absorptive functions. Pathogens, e.g., rotavirus, may strip the tips of the villi from large patches of the intestinal wall thus decreasing the surface area and decreasing by more than 50% the specific absorptive capacities of the intestine. The result is malabsorption which can cause malnutrition - most especially in a child already nutritionally compromised by repeated previous attacks of diarrhoea. Withholding food, even for one or two days, greatly exacerbates the malnutrition; this coupled with anorexia, caused partly by chronic potassium depletion, causes a vicious circle, i.e. diarrhoea causing malnutrition and malnutrition causing ever more frequent and severe diarrhoea. It is this diarrhoea/malnutrition cycle rather than acute dehydration that causes almost half of the five million deaths a year that are associated with diarrhoeal disease in children under five years old.
|