|
| |
|
Home > Diarrhoea > Consensus Statement of IAP National Task Force
|
Dialogue on Diarrhoea
Diarrheal Disease Messaging
Diarrhea Fact Sheet
Diarrhoea Management Training Course
Clinical Management of Acute Diarrhoea
IAP-Consensus Statement
Control in Bangladesh
Teaching Medical Students about Diarrhoeal Diseases
Consensus Statement of IAP National Task Force:
Status Report on Management of Acute Diarrhea
INDIAN PEDIATRICS
- VOLUME 41 - APRIL 17, 2004 - 335-348
Download this document in  pdf
376 kb pdf
376 kb
Statement
Consensus Statement of IAP National Task Force: Status Report on Management of Acute Diarrhea
Writing Committee:
Shinjini Bhatnagar
Nita Bhandari
U.C. Mouli
M.K. Bhan
Correspondence: Prof. M.K. Bhan* or Dr. Shinjini Bhatnagar,
Center for Diarrheal Disease and Nutrition Research, Department of
Pediatrics, All India Institute of Medical Sciences, New Delhi 110 029, India
E-mail: [email protected] ; [email protected]
* Convener |
Several important developments have been made in the field of management of
diarrhea in children as a result of research done in India and globally. It is
important to take follow-up steps to ensure that the benefits of new knowledge
reach affected children in India and at the same time ensure that new products
are not inappropriately used.
The Indian Academy of Pediatrics Committee For Framing Guidelines On The
Management Of Diarrhea In Children (members listed in Annexure 1) convened a
meeting at the All India Institute of Medical Sciences, New Delhi, to revise the
guidelines for management of diarrhea in children. The focus of this review was
oral rehydration solutions, zinc and probiotics in acute diarrhea, drug
treatment of dysentery, and management of diarrhea in the young infant and
severely malnourished subjects. The
meeting was convened to achieve a consensus on these issues based on careful
review of the literature and keeping in mind the requirements of treatment of
individual children as well as the needs of Diarrheal Diseases National Control
Program. As individual studies are often too small to yield precise estimation
of effect size, the recommendations are largely based on pooled data or
meta-analysis of randomized placebo controlled trials.
We summarize below the available data followed by consensus recommendations of
the group.
Reduced osmolarity ORS in acute diarrhea
The current standard WHO ORS has a sodium concentration of 90 mEq/L (glucose 110
mmol/L, osmolarity 311 mOsm/L), which corresponds to the stool electrolyte
composition in toxin-mediated diarrhea. However, it has worked well even in
young children with non-cholera diarrhea when used according to the recommended
guidelines with ready access to plain water during oral rehydration.
Several considerations lead to the clinical evaluation of reduced osmolarity
oral rehydration salts solutions and they have been examined by WHO(1).
Initially, one main concern was the potential risk of hypernatremia with
standard WHO-ORS in children with non-cholera diarrhea. There was also the
recognition that the standard WHO-ORS may provide too much sodium to edematous
children. In later years, there were reports of recurrent dehydration in young
infants treated with standard WHO-ORS on a weight to volume basis as replacement
of ongoing stool losses that was promptly reversed when
patients were kept nil orally and on intra-venous fluid regimens. Finally,
laboratory experiments showed that reduced osmolarity solutions (sodium 60 mmol/L,
glucose 80-120 mmol/L, osmolarity 240 mOsmol/L) promote water and sodium
absorption more efficiently than the WHO-ORS.
Review of clinical trials of reduced osmolarity oral rehydration salts solutions
Children with acute non-cholera diarrhea
A recently published meta-analysis of trials of reduced osmolarity ORS was
reviewed(2). The meta-analysis included all randomized trials in which a reduced
osmolarity ORS containing glucose, maltodextrin or sucrose (total osmolarity
210-268 mOsmol/L) and a sodium concentration ranging from 50 to 75 mEq/L was
used . These studies were conducted mainly in developing countries and included
well-nourished and malnourished children aged 1 month to 5 years with acute
diarrhea of duration <7 days with dehydration. Four of the studies were done in
India, two as part of large multi-center trials.
Results of the meta-analysis were as follows: (i) Use of reduced osmolarity ORS
was associated with a significant 39% reduction in need for IVF; need for IVF
was considered an important outcome measure as in many peripheral health
facilities, where IV therapy is often unavailable, reducing the need for
unscheduled IV therapy would reduce the risk of death from dehydration, (ii) 19%
reduction in stool output and (iii) 29% lower incidence of vomiting (Table I).
The incidence of hyponatremia (serum sodium <130 mEq/L) at 24 hours, evaluated
in 3 clinical trials was greater among children given reduced osmolarity ORS. 51
children treated with reduced osmolarity ORS and 36 children treated with
standard WHO ORS developed hyponatremia (OR = 1.45, 95% CI: 0.93 to 2.26). None
of these children were symptomatic. This difference was not statistically
significant but could be as much as twice that associated with standard WHO ORS.
TABLE I – Summary of The Results of the Published Meta-Analysis of All Randomized
Clinical Trials Comparing Reduced Osmolarity ORS With Standard WHO ORS in Children With Acute Non-Cholera Diarrhea
|
| Outcome |
Number of
studies reporting
|
Reduction in odds (95%CI) for children receiving reduced osmolarity ORS when compared to those receiving standard WHO ORS (311 mosmol/L) |
|
| Unscheduled IV
|
9 |
39% (19%, 53%) |
| Stool output |
12 |
19% (12%, 26%) |
| Vomiting |
6 |
29% (8%, 45%) |
|
Adapted from reference 1 and 2
|
Analysis of ORS efficacy stratified for sodium content
An analysis of all studies was conducted (1), stratifying them according to the
sodium content of the reduced osmolarity ORS: (i) reduced osmolarity ORS
containing less than 75 mEq/L of sodium (range 60 to 70 mEq/L), and (ii) reduced
osmolarity ORS containing exactly 75 mEq/L of sodium. Table II shows the
comparison of each of the two types of reduced osmolarity ORS with standard WHO
ORS and not a direct comparison with each other. ORS solution with a sodium
concentration of 75 mEq/L and sodium concentration of less than 75 mEq/L are
both more effective than standard WHO ORS with regard to need for unscheduled IV
therapy and occurrence of vomiting and that the incidence of hyponatremia, while
not significantly higher than for standard WHO ORS, could be up to double its
incidence. Although the effect size suggests a trend that is consistent with
greater reduction in stool output in the ORS with sodium concentration of less
than 75 mEq/L, the test for interaction could not differentiate between the
efficacy of ORS solution with a sodium concentration of 75 mEq/L and and that of
ORS solution containing sodium less than 75 mEq/L, even on unidirectional tests
of significance.
Children with cholera diarrhea
In the pooled data (1) of all studies with cholera diarrhea in children there
was a small, but statistically significant reduction, in mean serum sodium at 24
hours in patients receiving reduced osmolarity ORS (sodium 70-75 mEq/ L, glucose
75-90 mmol/L, osmolarity 245-268 mOsm/L) when compared with those given standard
WHO ORS [(mean difference 0.8 mEq/L, 95% CI: 0.6 to 1.0). The children receiving
reduced osmolarity ORS did not have a higher risk, than those receiving standard
WHO ORS, of developing hyponatremia (serum sodium <130 mEq/L) at 24 hours (RR =
1.8, 95% CI: 0.9 to 3.2), but a possible doubling of the incidence cannot be
ruled out based on the confidence intervals. None of these children with
hyponatremia were symptomatic. Stool output at 24-hours was not different
between treatment groups in children with cholera in the multicentre study
(sodium 75 mEq/L, glucose 75 mmol/L, osmolarity 245 mOsm/L). In the other two
studies, however, stool output was reduced by
about 30% in children with cholera who were treated with reduced osmolarity ORS.
|
TABLE II–Pooled Analysis Stratified According to the Sodium Content of the
Reduced Osmolarity ORS
|
|
|
Reduced OSM ORS with < 75 mEq/L of sodium in comparison to standard WHO ORS |
Reduced OSM ORS with 75 mEq/L of sodium in comparison to standard WHO ORS |
|
|
|
N = 4 studies
|
N = 4 studies
|
Odds ratio (95% CI) for unscheduled IV therapy
for patients given RED OSM ORS when
compared to those given standard WHO ORS
|
N = 678 children
0.65 (0.41 to 1.00) |
N = 1175 children
0.56 (0.39 to 0.80) |
Ratio of geometric means (95%CI) for stool
output in children given RED OSM ORS when compared to those given standard WHO ORS
|
N = 8 studies
N = 771 children
0.69 (0.49 to 0.98) |
N = 4 studies
N = 1049 children
0.88 (0.71 to 1.06) |
Odds ratio (95%CI) for vomiting for patients
given RED OSM ORS when compared to those
given standard WHO ORS
|
N =3 studies
N = 270 children
0.49 (0.27 to 0.91) |
N = 3 studies
N = 1031 children
0.74 (0.58 to 0.95) |
|
Odds ratio (95%CI) for hyponatremia (<130
mEq/L) for patients given RED OSM ORS when
compared to those given standard WHO ORS |
N = 3 studies
N = 139 children
Not analyzed |
N = 3 studies
N = 1120 children
1.45 (0.93 to 2.26) |
|
|
Reproduced from Reference 1 |
|
Reduced osmolarity ORS in adults with cholera
The combined analysis of three studies(1) that compared the efficacy and safety
of reduced osmolarity ORS (osmolarity 245-249 mOsm/L) to that of standard WHO
ORS in adults with cholera showed a minimal, and statistically insignificant,
mean reduction of
0.5 ml/kg (95% CI: –14.6 to +15.6) in stool output during the first 24 hours
among patients given reduced osmolarity ORS. A small, but statistically
significant reduction in mean serum sodium of 1.3 mEq/L (95% CI: 0.3 to 2.3) was
observed at 24-hours in patients treated with reduced osmolarity ORS when
compared to those given standard WHO ORS. None of these patients who developed
hyponatremia became symptomatic.
Recommendations by the WHO Task Force, New York, July 2001
The WHO Meeting of Experts(1) concluded that there are programmatic and logistic
advantages of using a single solution around the world for all causes of
diarrhea in all ages. After reviewing the data the group of experts proposed
that reduced osmolarity ORS with 75 mEq/L of sodium and 75 mmol/ L of glucose is
effective in adults and children with cholera and that reduced osmolarity ORS
solution with 60 mEq/L of sodium does not seem to be significantly better than
reduced osmolarity ORS solution containing 75 mEq/L of sodium. They concluded
that safety data in patients with cholera, while limited, are reassuring.
The WHO Meeting of Experts(1) further recommended that this formulation falls
within the ranges defined by the WHO’s Program for the Control of Diarrheal
Diseases
(CDD) in March 1992 for a safe and efficacious oral rehydration solution, which,
therefore, remain unchanged. The recommended ranges were that the total
substance concentration (including that contributed by glucose) should be within
the range 200-311 mmol/L. The individual substance concentration of glucose
should at least equal that of sodium, but should not exceed 111 mmol/L and that
of sodium should be within the range of 60-90 mmol/L. The concentrations of
potassium, citrate and chloride should be with-in the range of 15-25 mmol/L,
8-12 mmol/L and 50-80 mmol/L respectively as shown below
(1).
Recommendations of the IAP National Task Force for use of ORS in diarrhea,
August 18-19, 2003
- All doctors should prescribe ORS for all ages in all types of diarrhea.
- The group noted that the new improved universal ORS recommended by the WHO
containing sodium 75 mmol/L and glucose 75 mmol/L, osmolarity 245 mOsmol/L is
acceptable for all ages and may be made freely available by the Government.
However it was proposed that a pediatric ORS containing sodium 60 mmol/L,
glucose 84 mmol/L, osmolarity 224
mOsmol/L is the most suitable solution for children and the industry should be
encouraged to produce such a formulation.
- The current formulations ORS A and ORS citrate allowed in the Indian Pharmacopia,
1996(2) should no longer be used and only the above recommended formulations be
in the market. Consideration should be given to a different color code for the
two formulations so that the formulation containing sodium 60 mmol/L, glucose 84
mmol/L, osmolarity 224 mOsmol/L is identified as more suitable for children.
This can be further symbolized by sporting a child’s picture.
- The powder packet to make 1 liter of solution should be continued. Since mothers
tend to use ORS a glass at a time, a measuring device should be included inside
to measure the required amount of powder accurately for 200 ml of fluid.
- The group was deeply concerned that ORS was not available free of cost at public
institutions. It recommended that measures should be taken by the Government to
improve its availability and reduce its cost.
- The group did not currently recommend marketing of ORS with additives
(probiotics, minerals). They should only be permitted after demonstrating
benefit in studies carried out in Indian patients as breastfeeding rates,
dietary habits and intestinal flora varies from European and North American
children.
Zinc in the Treatment of Acute Diarrhea
The rationale for use of specific nutrients as treatment of acute diarrhea is
based on their effects on immune function or on intestinal structure or function
and on the epithelial recovery process during diarrhea.
Zinc deficiency has been found to be widespread among children in developing
countries, and occurs in most of Latin America, Africa, the Middle East and
South Asia. Zinc has been identified to play a critical role in metallo-enzymes,
polyribosomes, the cell membrane, and cellular function, leading to the belief
that it also plays a central role in cellular growth and in the function of the
immune system. Intestinal zinc losses during diarrhea aggravate pre-existing
zinc deficiency. Convincing evidence for its clinical importance has come from
recent randomized controlled trials of zinc during acute diarrhea.
Clinical efficacy of zinc as an adjunct to oral rehydration therapy in acute
diarrhea
The results of pooled analyses (3) of zinc treatment trials in children with
acute diarrhea and the findings of subsequent studies are summarized in Table
III. The main features of these trials include the randomized placebo controlled
design subjects’ aged between 6 months and 3 years, and daily elemental zinc
dose ranging from 10 to 30 mg per day.
|
TABLE III – Results of Pooled-Analysis and Subsequent Randomized Controlled Trials
in Children with Acute Diarrhea Comparing Impact of Zinc with that of Placebo.
|
Study |
Number of subjects |
Effect size (95%CI) |
|
Risk of continuation of diarrhea
|
|
Relative hazards
|
|
Pooled analysis (3) |
1252/1194 |
0.85 (0.76 to 0.95) |
Subsequent studies in South-East Asia |
|
|
|
Strand et al. (4) |
442/449 |
0.79 (0.68 to 0.93) |
|
Bahl et al. (5) |
404/401 |
0.89 (0.80 to 0.99) |
|
Bhatnagar et al. (6) |
132/134 |
0.76 (0.59 to 0.97) |
Diarrhea lasting >7 days
|
|
Odds ratio
|
|
Pooled analysis |
1252/1194 |
0.78 (0.56 to 1.09) |
Subsequent studies in South-East Asia
|
|
|
|
Strand et al. |
442/449 |
0.57 (0.38 to 0.86) |
|
Bahl et al. |
404/401 |
0.61 (0.33 to 1.12) |
|
Bhatnagar et al. |
132/134 |
0.09 (0.01 to 0.73) |
Stool output
|
|
Difference in means
or Ratio of geometric means |
|
Roy et al. (7) |
57/54 |
–91 g |
|
Dutta et al. (8) |
44/36 |
–900 g (–1200 to –590) |
|
Bhatnagar et al. |
132/134 |
0.69 g/kg (0.48, 0.99) |
|
|
Reproduced from Reference 1. |
|
In the trials subjected to pooled analysis, zinc supplemented children had 16%
faster recovery (95% CI 6% to 22%). Zinc treatment also resulted in a 20%
reduction (95% CI –2%
to 38%) in the odds of acute episodes lasting >7 days. The findings of the
subsequent trials are consistent with the conclusions of the meta analysis. The
study by Bhatnagar et al. is of interest as it was hospital based, involved
cases of acute diarrhea with dehydration and measured impact on stool output. In
the zinc treated children, the total stool output was reduced by 31% (95% CI 1%
to 52%) than in the placebo group.
The effect of zinc did not vary significantly with age, or nutritional status
assessed by anthropometry. The effects were not dependent upon the type of zinc
salts: zinc sulfate, zinc acetate or zinc gluconate. The optimal dose is yet to
be determined but there seems to be little gain in efficacy when the commonly
used 20 mg daily dose of elemental zinc was increased to 30-40 mg daily.
Majority of the studies so far were conducted in South East Asia, where zinc
deficiency is common. Finally, there is relatively little data on children aged
less than 6 months to allow any conclusions about efficacy in this age group.
Another study conducted in Bangladesh(9) used a cluster randomized design to
evaluate the effect on mortality and morbidity of providing daily zinc for 14
days to children with diarrhea as part of the diarrhea treatment programme in
the community. The intervention and the comparison clusters were both given ORS
and advice on feeding during diarrhea. The children in the zinc cluster had a
shorter duration (hazard ratio 0.76, 95%CI
0.65 to 0.90) and lower incidence of diarrhea (rate ratio 0.85, 95% CI 0.76 to
0.96) than children in the comparison group, lesser
admission to hospital of children with diarrhea (rate ratio 0.76; 95% CI 0.59 to
0.98), and lower mortality due to non injury deaths, notably diarrhea or
pneumonia (rate ratio 0.49; 95% CI 0.25 to 0.94) in the zinc treated cluster.
The data are consistent in showing a beneficial effect of zinc in acute
diarrhea.
Zinc fortified ORS
The efficacy of 40mg elemental zinc mixed with a liter of standard WHO ORS
solution was compared with ORS without zinc and with zinc syrup administered
separately from ORS(5). While zinc-ORS was superior to ORS alone, it was less
efficacious in reducing duration of the episode than zinc supplements given
separately from the ORS solution. The data are currently too limited.
The therapeutic benefits in acute diarrhea may be attributed to effects of zinc
on various components of the immune system and its direct gastrointestinal
effects. Zinc deficiency is associated with lymphoid atrophy, decreased
cutaneous delayed hypersensitivity responses, lower thymic hormone activity, a
decreased number of antibody forming cells and impaired T killer cell activity.
Zinc deficiency has also been recently shown to affect the differentiation of
CD4 response towards Th1 rather than Th2 pathway. The direct intestinal effects
of zinc deficiency include decreased brush border activity, enhanced secretory
response to cholera toxin, and altered intestinal permeability, which is
reversed by supplementation.
WHO constituted a Task Force consisting of a group of experts, which met in New
Delhi in May 2001(10). They reviewed all the studies done till 2001 and
concluded that:
- Zinc supplementation, given at a dose of about 2 RDA per day (10 to 20 mg per
day) for 14 days, is efficacious in significantly reducing severity of diarrhea
as well as duration of the episode.
- They recommended effectiveness studies to assess different strategies for
delivering zinc supplementation to children with diarrhea. These studies should
investigate the feasibility, sustainability and cost effectiveness of different
zinc delivery mechanisms, and monitor variables such as ORS solution
consumption, antibiotic use rate, non diarrhea morbidity and overall mortality.
They recommended further research to determine the effect of zinc
supplementation in young infants.
Recommendations of the IAP National Task Force for use of Zinc in Diarrhea,
August 18-19, 2003
- Based on studies in India and other developing countries there is sufficient
evidence to recommend zinc in the treatment of acute diarrhea as adjunct to oral
rehydration. However, ORS remains the mainstay of therapy during acute diarrhea
and zinc has an additional modest benefit in the reduction of stool volume and
duration of diarrhea as an adjunct to ORS. Under all circum-stances, oral
rehydration therapy must remain the main stay of treatment.
- Treatment of acute diarrhea with zinc may have benefits on morbidity and
mortality from other childhood infections and these should be further
investigated.
- A uniform dose of 20 mg of elemental zinc should be given during the period of
diarrhea and for 7 days after cessation of diarrhea to children older than 3
months. Re-commendations for below 3 months must await further research.
- Based on all the studies the group proposed that zinc salts e.g., sulphate,
gluconate or acetate may be recommended.
- The industry should be encouraged to prepare a zinc formulation, which
contains only zinc. Until these are available, the group proposed that
formulations providing vitamins together with zinc may be used provided doses of
former are within 1 RDA. Iron containing formulations should not be used with
zinc as iron interferes with zinc absorption.
Probiotics in the Treatment of Diarrhea
Probiotics are nonpathogenic micro-organisms that, when ingested exert a
positive influence on the health or physiology of the host. They consist of
either yeast or bacteria, predominantly Bifidobacterium and Lactobacillus. There
is some preliminary evidence that ingestion of probiotics offers therapeutic
benefit in the treatment of acute gastroenteritis in children. There are several
possible mechanisms by which probiotics may exert their clinical effects. They
can protect the intestine by competing with pathogens for attachment,
strengthening tight junctions between enterocytes and enhancing the mucosal
immune response to pathogens.
The group reviewed the available published randomized controlled studies on
therapeutic benefits of probiotics in acute diarrhea. Most of the studies are
small in size except for a single large multicentre trial and have been done in
developed countries. None of the studies are from India. Some inferences are
possible from the recently published meta-analyses.
Results of meta-analysis of RCTs comparing probiotics with placebo in
hospitalized children aged 1-48 months with diarrhea <7 days
Overall effect size of probiotics on diarrheal duration
The first meta-analysis reported details of
10 treatment studies involving hospitalized children aged 1-48 months with
diarrhea less than 7 days(11). All studies were conducted in developed countries
from the West except for one from Thailand. The probiotics used were
Lactobacillus GG, Lactobacillus reuteri, Saccharomyces boulardii, Streptococcus
thermophillus lactis, Lactobacillus acidophilus and Lactobacillus bulgaricus.
Rota-virus was the cause of diarrhea in more than 75% cases in studies from
Finland, 28% in Russia, 48% in Thailand and 35% in the multicentre trial. Figure
1 summarizes the pooled effect of probiotics on duration of diarrhea in 7 trials
involving 679 children. Probiotics significantly reduced the duration of
diarrhea compared with the placebo by 21 hours (the pooled weighted mean
difference assuming the random–effect model was –20.1 (95% CI –26.1, –14.2).
The second meta-analysis included most of the studies from the first analysis
and one small study from Pakistan, which enrolled only 36 subjects(12). The
results were consistent with the earlier meta-analysis reporting a reduction in
diarrheal duration of
0.7 days (95% CI 0.3 to 1.2) in subjects whoreceived lactobacillus compared with
those who received the placebo.
Effect size by type of Lactobacillus strains
The first meta-analysis(11) further reported subgroup analysis for different
probiotic strains (Fig. 1). Both LGG (pooled weighted mean difference assuming
the random-effect model was –22 (95% CI –31.3, –21.8) and L. reuteri (pooled
weighted mean difference assuming the random–effect model was –25.3 (95% CI–40.7
to –9.95) significantly reduced the duration of diarrhea as compared to the
placebo. There was only one study with Lactobacillus acidophilus which reported
a trend in the reduction of diarrheal
duration but this was not statistically significant –13.6 (95% CI –28 to 0.83) .
Only LGG (data not shown) showed a consistent effect on the reduction in risk of
diarrhea lasting >3 days (pooled estimate RR 0.4 95% CI 3 to 9).
|
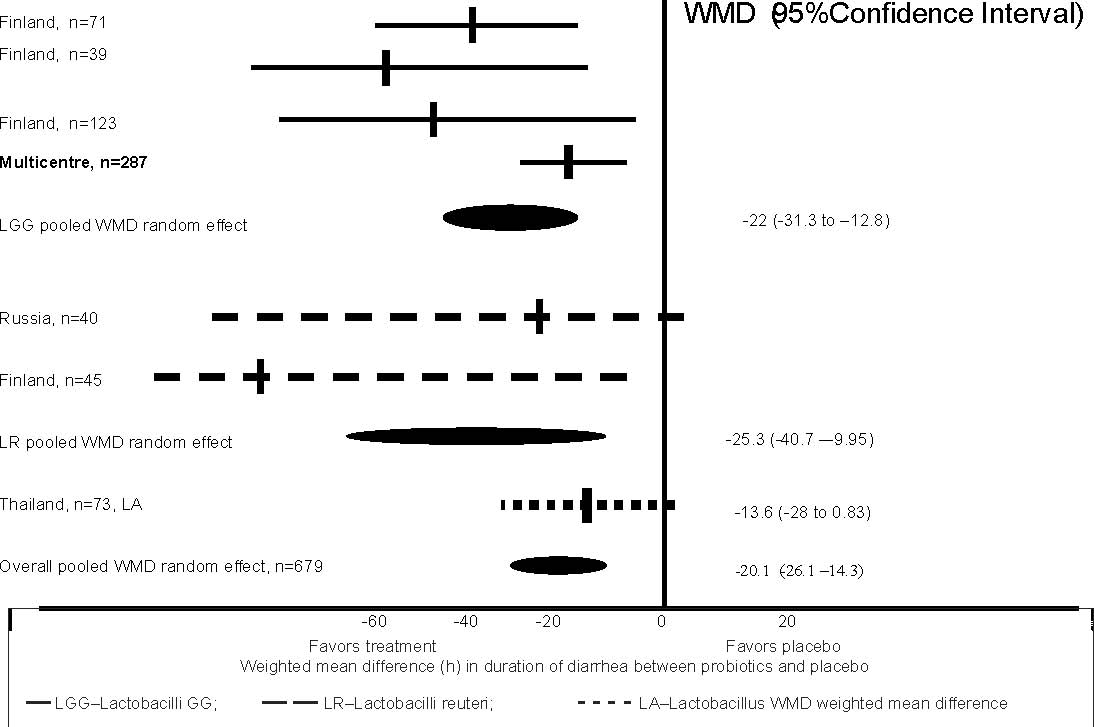
Fig. 1. Results of meta-analysis of RTCs comparing problems with placebo in
hospitalized children aged 1-48 months with diarrhea <7 d.
Adapted from Reference 12.
Effect size by different types of Lactobacillus strains on types of diarrhea;
viral or invasive
LGG and L. reuteri significantly reduced duration of diarrhea as compared with
the placebo in 297 children with rotavirus diarrhea (weighted mean difference
–24.8h (95% CI –31.8 to –17.9) while a similar effect was notseen in a small sub-group of
subjects with invasive diarrhea 1.3h (95%CI –15.3 to 17.9) (Fig. 2).
|
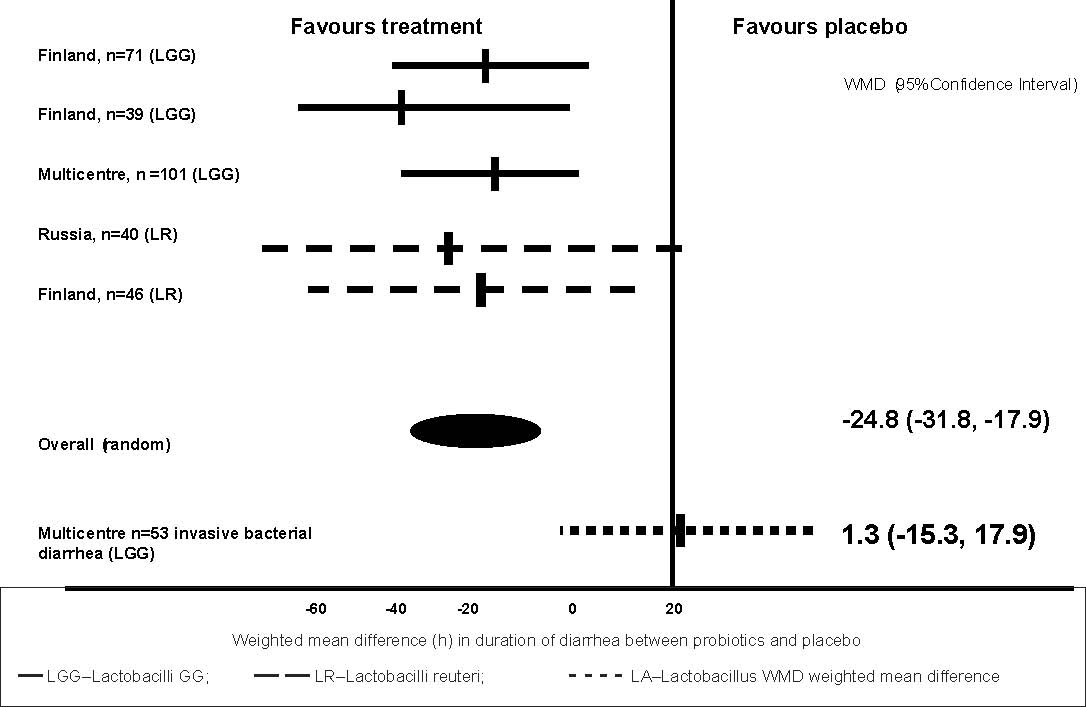
Fig. 2.
Efficacy of probiotics in reducing duration of rotavirus diarrhea.
Adapted from Reference 12.
Probiotics available in the Indian market
Lactobacillus GG is not available in the Indian market. The other commonly
available probiotics are Lactic Acid bacillus, Lactobacillus acidophilus and Saccharomyces boulardii.
Recommendations of the IAP National Task Force for Use of Probiotics in
Diarrhea, August 18-19, 2003
The group recommended that based on the above studies there is presently
insufficient evidence to recommend probiotics in the treatment of acute diarrhea
in our settings as:
- Almost all the studies till now were done in developed countries except for
one very small study from Pakistan. It may not be possible to extrapolate the
findings of these
studies to our setting where the breast feeding rates are high and the microbial
colonization of the gut is different.
- The effect of probiotics is strain related and there is paucity of data to
establish the efficacy of the probiotic species (namely L. acidophilus, Lactic
Acid Bacteria) available in the Indian market. To recommend a particular species
it will have to be first evaluated in randomized controlled trials in Indian
children.
- The earlier studies have documented a beneficial effect on rotavirus diarrhea
which was present in >75% of cases in studies from the west. Rotavirus
constitutes about 25% of diarrhea in hospitalized children and 15% in outpatient
practice in India.
- The primary outcome analyzed in all the studies was the duration of diarrhea.
The more objective parameter of stool output was not evaluated.
Treatment of Acute Diarrhea in the Young Infant (< 2 months)
The IAP group suggested that for assessment of diarrhea in young infants (up to
age 2 months) recommendations by the Integrated Management of Neonatal and
Childhood Illnesses, which is an adapted version of Integrated Management of
Child-hood Illnesses for India, should be followed. The following additional
recommendations were made:
- Infants who are breastfed and have no dehydration do not need ORS and mothers
should be advised to increase breast feeds. Young infants with de-hydration
should be treated as has been recommended for other children with dehydration.
- Low osmolarity ORS (Pediatric ORS of glucose 80 mmol/L, sodium 60 mmol/L and
osmolarity 224 mOsmol/L) should be used. If this ORS is not available the new
WHO recommended ORS (glucose 75 mmol/L,
sodium 75 mmol/L and osmolarity 245 mOsmol/L)
may be used. There is no need to give extra plain
water during rehydration with ORS.
- 3. Third generation cephalosporins (intravenous ceftriaxone, cefotaxime, oral
cefixime) or intravenous ciprofloxacin
should be given for treatment of dysentery. Where hospitalization is not
possible, the drugs can be used orally.
Antibiotic Use in Acute Dysentery
The issue of indiscriminate use of antibiotics as well the increasing
incidence of antibiotic resistance is causing great concern.
The national diarrheal disease control program
currently recommends the use of
cotrimoxazole as the first line drug for the management of acute bloody
diarrhea.
Recent studies from India and other Asian, as well as non-Asian developing
nations have shown
high rates of resistance of shigella to cotrimoxazole, ampicillin, chloramphenicol
and tetracyclines. Resistance rates to nalidixic acid and quinolones are
still low.
The following consensus was reached on the issues of antibiotics:
- Antibiotics are indicated only for acute bloody diarrhea.
- Antibiotics are not indicated for children with acute diarrhea and
no visible blood in stools, with pus cells on stool microscopy because
of poor specificity of the test. Routine stool examination or stool
cultures have no useful role in the management of usual cases of acute
bloody diarrhea.
- Data on resistance of shigella and other enteric pathogens to
antibiotics is still limited and is inadequate to make a uniform single
recommendation for the entire country. Therefore, a concerted attempt
needs to be made to produce data regarding resistance
patterns from all over the country.
- In areas where resistance rates to cotrimoxazole exceed 30%, nalidixic acid should be used as the first line drug for
the treatment of acute bloody diarrhea. In case of poor response, norfloxacin, ciprofloxacin or a third generation cephalosporin must be used as second and third line drugs.
- In areas where such data does not exist or rates of resistance have been demonstrated to be lower than 30%, cotrimoxazole should be the first line drug to
manage acute bloody diarrhea in all but
high-risk cases; these include infants who have not
been breastfed and severely
malnourished children. In these
high-risk groups nalidixic acid or norfloxacin should be the first line
drug.
- Entamoeba histolytica and
helminths rarely ever cause acute diarrhea
in children. Metronidazole and antihelminthics therefore have no role in the routine management of acute bloody diarrhea. Metronidazole should be used when cases of acute
dysentery fail to respond to second line drugs for dysentery such
as norfloxacin or when a stool examination
has confirmed trophozoites of Entamoeba hystolitica.
- Aminoglycosides like gentamicin and amikacin have a
poor spectrum of activity against shigella species and therefore
they are ineffective in the management of acute bloody diarrhea.
- Antibiotic Use in Acute Dysentery
(Table IV).
Antiemetics in Acute Diarrhea
Vomiting is the commonest symptom associated with acute diarrhea in children. Often vomiting is particularly
distressing to the parents and
therefore, antiemetics are frequently
prescribed. Concerns were raised by
members, on their use, in view of the serious side effects these drugs can produce. Low osmolarity ORS is expected to reduce the incidence of vomiting in children with acute
gastroenteritis. Most children with vomiting can be managed with frequent small
sips (5-10 ml) of ORS.
- Antiemetics should be reserved for children in whom the vomiting is severe,
recurrent and interferes with ORS intake.
- Among the available antiemetics in use for children domperidone is the safest
with no central nervous system side effects. Therefore. the group recommended a
single dose of domperidone in children with severe vomiting. Continued use is
not recommended. Domperidone should be used at a dose of 0.1-0.3 mg/kg/dose.
- In view of serious side effects metoclopramide is not recommended for the
management of vomiting in acute gastroenteritis.
Management of a HIV Positive Patient with Acute Diarrhea
In children who are HIV positive or are immunocompromised due to other
immunodeficiency states the management of acute diarrhea varies from the
management in the immunocompetent. The causative organisms of diarrhea differ
and the consequences of diarrhea are more severe in these children, particularly
in those with Cryptosporidiosis(13). The primary determinant of the organisms
and consequences is whether the child is receiving antiretroviral therapy and
its intensity. A HIV positive child with acute diarrhea must have a stool
culture, stool examined for ova, cysts and atypical protozoa (includes isospora,
microspora and cryptospora on at least three occasions) and CD4 counts
determined.
Children with optimum antiretroviral therapy and CD4 counts >500
Children on intense retroviral therapy and CD4 counts greater than 500 can be
managed like normal children with diarrhea.
Children on inadequate or no antiretro-viral therapy or CD4 counts <500
- Children with low CD4 counts (<500) or not on antiretroviral drugs and those
children who have failed to respond to standard first line therapy of acute
diarrhea must be started on a combination of ciprofloxacin and metronidazole
along with adequate amounts of ORS.
- In cases where the response is poor despite 5-7 days of therapy repeat stool
examination for ova, cysts and atypical protozoa (includes isospora, microspora
and cryptospora on at least 3 occasions) should be done and oral cotrimoxazole
should be added.
- Children who do not respond to the above therapy by a week should be referred to
a higher center for investigations and treatment. These include flexible
sigmoido-scopy with a biopsy of mucosa for typical pathological changes as in
cytomegalo-virus and culture of rectal tissue for bacteria (especially campylobacter).
- 4. Oral Nitazoxanide (200 mg doses in children aged 4-11 years and 100-mg doses
in children aged 1-3 years given in two divided doses for a total of three
days), azithromycin (10 mg/kg/ once daily for 10 days), clarithromycin (15
mg/kg/day in two divided doses) or paramomycin can be tried for management of
Cryptosporidium. Gancyclovir (12 mg/kg in two doses per day IV) is recommended
for 6 weeks when pathological evidence of Cytomegalovirus infection is
established.
|
TABLE IV – Antibiotic use in Acute Dysentery.
|
S.No. |
Drug |
Dose/Kg body wt/ day |
Divided doses |
Duration (days) |
|
|
1 |
Nalidixic acid |
55 mg/kg / day |
3 |
5 |
|
2 |
Norfloxacin |
20 mg /kg /day |
2 |
5 |
|
3 |
Cotrimoxazole |
5-8mg/kg/day |
2 |
5 |
|
4 |
Ciprofloxacin |
< 20 kg: 125 mg |
2 |
5 |
|
|
|
> 20 kg: 250 mg |
2 |
5 |
|
|
Acknowledgements
We acknowledge Ministry of Health, Government of India and the WHO for providing
technical support and CMS for their support for the meeting.
REFERENCES
- Reduced osmolarity oral rehydration salts (ORS) formulation. A report from a
meeting of experts jointly organized by UNICEF and WHO. UNICEF HOUSE, New York,
USA, 18 July, 2001. WHO/FCH/CAH/0.1.22
- Hahn SK, Kim YJ, Garner P. Reduced osmolarity oral rehydration solution for
treating dehydration due to diarrhea in children: systematic review. BMJ, 2001;
323 : 81-85.
- Zinc Investigators’ Collaborative Group. Bhutta ZA, Bird SM, Black RE, Brown KH,
Gardner JM, Hidayat A et al. Therapeutic effects of oral zinc in acute and
persistent diarrhea in children in developing countries: pooled analysis of
randomized controlled trials. Am J Clin Nutr 2000; 72: 1516-1522.
- Strand TA, Chandyo RK, Bahl R, Sharma PR, Adhikari RK, Bhandari N, et al.
Effectiveness and efficacy of zinc for the treatment of acute diarrhea in young
children. Pediatrics 2002; 109: 898-903.
- Bahl R, Bhandari N, Saksena M, Strand T, Kumar G.T, Bhan MK et al. Efficacy of
zinc fortified oral rehydration solution in 6-35 month old children with acute
diarrhea. J Pediatr 2002; 141: 677-682.
- Bhatnagar S, Bahl R, Sharma PK, Kumar GK, Saxena SK, Bhan MK. Zinc treatment
with oral rehydration therapy reduces stool output and duration of diarrhea in
hospitalized children; a randomized controlled trial. J Pediatr Gastroenterol
Nutr 2004; 38: 34-40.
- Roy SK, Tomkins AM, Akramuzzaman SM, Behrens RH, Haider R, Mahalanabis D et al.
Randomized controlled trial of zinc supple-mentation in malnourished Bangladeshi
children with acute diarrhea. Arch Dis Child 1997; 77: 196-200.
- Dutta P, Mitra U, Datta A, Niyogi SK, Dutta S, Manna B et al. Impact of zinc
supplementation in malnourished children with acute diarrhea. J Trop Pediatr
2000; 46: 259-263.
- Baqui AH, Black RE, El Arifeen S, Yunus M, Chakraborty J, Ahmed S et al. Effect
of zinc supplementation started during diarrhea on morbidity and mortality in
Bangladeshi children: Community randomized trial. BMJ 2002; 325(7372): 1059.
- Effect of zinc supplementation on clinical course of acute diarrhea. Report of a
Meeting, New Delhi, 7-8 May 2001. J Health Popul Nutr 2001;19: 338-346.
- Szajewska H, Mrukowicz JZ. Probiotics in the treatment and prevention of acute
infectious diarrhea in infants and children: a systematic review of published
randomized, double-blind, placebo-controlled trials. J Pediatr Gastroenterol
Nutr. 2001; 33 Suppl 2: S17-25.
- Van Niel CW, Feudtner CF, Garrison MM, Christakis DA. Lactobacillus therapy for
acute infectious diarrhea in children: A meta-analysis. Pediatrics 2002; 109:
678-684.
- Kosek M, Alcantara C, Lima AA, Guerrant RL. Cryptosporidiosis: An update. Lancet Infect Dis 2001; 1: 262-269.
Annexure - I
List of Participants (alphabetically)
Dr. Arun Kumar Agrawal
Secretary, Indian Academy of Pediatrics
subspecialty Chapter of Community Pediatrics
Dr. N. K. Arora
All India Institute of Medical Sciences, New Delhi
Dr. S. C. Arya
Sir Ganga Ram Hospital, New Delhi
Dr. C. R. Banapurmath
National Convener,
Breastfeeding and Lactation Management Committee,
Indian Academy of Pediatrics
Dr. Ashish Bavdekar
Secretary, Indian Academy of Pediatrics
subspecialty Chapter of Gastroenterology
Dr. M. K. Bhan (Convener)
All India Institute of Medical Sciences, New Delhi
Dr. Nita Bhandari
All India Institute of Medical Sciences, New Delhi
Dr. Shinjini Bhatnagar
All India Institute of Medical Sciences, New Delhi
Dr. Swati Bhave
Indraprastha Apollo Hospital and MAX Health Care,
New Delhi
Dr. Panna Choudhury
Chairperson, Indian Academy of Pediatrics
subspecialty Chapter of Nutrition
Dr. A. P. Dubey
Secretary, Indian Academy of Pediatrics
subspecialty Chapter of Nutrition
Dr. T. K. Ghosh
Secretary, Indian Academy of Pediatrics
subspecialty Chapter of Infectious Diseases
Dr. G. S. Hathi
Chairperson, Breastfeeding and Lactation Management Committee,
Indian Academy of Pediatrics
Dr. B. Kishore (Special Invitee)
Ministry of Health & Family Welfare, New Delhi
Dr. S. K. Mittal
Maulana Azad Medical College, New Delhi
Dr. U. C. Mouli
All India Institute of Medical Sciences, New Delhi
Dr. D. Mukherjee
President, Indian Academy of Pediatrics 2002
Dr. M. K. C. Nair
President, Indian Academy of Pediatrics 2004
(could not attend)
Dr. Sushma Narayan
Municipal Corporation of Delhi
Dr. A. K. Patwari
WHO India Country Office, New Delhi
Dr. S. Sarkar (Special Invitee)
Ministry of Health & Family Welfare,
New Delhi
(could not attend)
Dr. H. P. S. Sachdev
President, Indian Academy of Pediatrics 2003
Dr. R. C. Shah
Chairperson, Indian Academy of Pediatrics
subspecialty Chapter of Infectious Diseases
Dr. Mamta Sharma
Deen Dayal Upadhayaya Hospital,
New Delhi
Dr. N. K. Shah
Hony. Secretary General Indian Academy of Pediatrics
(could not attend)
Dr. Surjit Singh
Convenor, Immunization Committee of
Indian Academy of Pediatrics
Dr. A. D. Tewari
Chairperson, Indian Academy of Pediatrics
subspecialty Chapter of Community Pediatrics
Dr. S.K. Yachha
Chairperson, Indian Academy of Pediatrics
subspecialty Chapter of Gastroenterology
(1) WHO
recommended range for safe and efficacious oral rehydration solution
The total substance concentration should be within the range 200-311 mmol/L (including that contributed by glucose)
The individual substance concentration of:
| Glucose |
should at least equal that of sodium, but should not exceed 111 mmol/L |
| Sodium |
should be within the range of 60-90 mmol/L |
| Potassium |
should be within the range of 15-25 mmol/L |
| Citrate |
should be within the range 8-12 mmol/L |
| Chloride |
should be within the range 50-80 mmol/L |
(2)The Indian Pharmacopia (IP) and ORS recommendations:
The two ORS formulations in the IP, 1996 are:
| |
ORS-A
|
ORS-Citrate (the current WHO formulation) |
| Sodium chloride |
3.5 g |
3.5 g |
| Potassium chloride |
1.5 g |
1.5 g |
| Sodium citrate |
2.9 g |
2.9 g |
| Anhydrous dextrose |
27 g |
20 g |
| or |
|
|
| Dextrose monohydrate |
29.7 |
20 g |
ORS-A contains glucose in very high concentration. |
|
updated: 7 May, 2015
|

