|
| |
Home > RotaVirus Disease and Vaccines |
Rotavirus Disease and Vaccines On-line Resources
Worldwide, almost every child will have at least one rotavirus infection before
he or she is five years old. The virus is so contagious and resilient that
providing clean water and promoting proper hygiene do not significantly reduce
incidence, which is nearly the same in industrialized and developing countries.
Additionally, because rotavirus usually causes profuse vomiting, ORS/ORT is
difficult to administer. Rotaviruses are a genus of viruses belonging to the Reoviridae family. Seven
major groups have been identified, three of which (groups A, B, and C) infect
humans, with group A being the most common and widespread one.
Rotavirus disease
 |
- Rotavirus (pronounced "row-tuh-virus") is the most common cause of severe gastroenteritis in children worldwide.
- Rotavirus is responsible for the deaths of an estimated 600,000 children each year, 80 percent of whom live in developing countries.
- Rotavirus is found in all countries. Most children have had one or more rotavirus infections by the age of 5.
- In young children, rotavirus disease is characterized by diarrhea, vomiting, fever, and severe dehydration. Death is caused by dehydration due to rotavirus infection, not by the virus itself.
- Rotavirus disease cannot be treated with antibiotics or other drugs. Regardless of hygiene practices or access to clean water, nearly every child
in the world will be infected with rotavirus before age 5. Vaccination is the only viable measure to prevent severe rotavirus illness.
|
Rotavirus vaccines
- Studies of two new rotavirus vaccines recently demonstrated their safety and efficacy among children in middle- and high-income countries.
- Clinical trials have been launched, and additional studies are planned, to evaluate the impact of vaccines as a method for the prevention of severe
rotavirus disease in developing countries. Results generated from these trials will help national governments make informed decisions about
introducing the vaccines into the public sector.
- Enhancing diarrheal disease control through a combined prevention and treatment strategy—incorporating rotavirus vaccine; new, low-osmolarity
formulations of oral rehydration solution; and zinc supplementation during diarrhea episodes—can rapidly and significantly reduce child mortality where
diarrheal disease is a serious burden.
|
 The
10th International Rotavirus Symposium will be held 19-21 September, 2012 in
Bangkok, Thailand. It will bring together interested stakeholders to provide
an update on new data and relevant research that will inform public health
agendas related to prevention of rotavirus gastroenteritis. Main Session Topics: Participants will discuss the latest results of trials of new rotavirus
vaccines in developing country settings, issues in vaccine policy and
introduction, and early post-marketing data on vaccine impact and safety. The
10th International Rotavirus Symposium will be held 19-21 September, 2012 in
Bangkok, Thailand. It will bring together interested stakeholders to provide
an update on new data and relevant research that will inform public health
agendas related to prevention of rotavirus gastroenteritis. Main Session Topics: Participants will discuss the latest results of trials of new rotavirus
vaccines in developing country settings, issues in vaccine policy and
introduction, and early post-marketing data on vaccine impact and safety. |
|
27 September, 2011
GAVI approves rotavirus vaccine funding for 16 new countries, 12 in Africa Until today, life-saving rotavirus vaccines were not accessible for most
children in Africa, the continent with a staggering burden of rotavirus
disease and where vaccines are desperately needed to prevent severe
rotavirus diarrhea and save children’s lives: • Where nearly a quarter of a million children die of rotavirus disease each year.
• Where roughly 40% of children hospitalized for severe diarrhea have rotavirus.
• Where urgent care and treatment for severe rotavirus diarrhea is often limited or unavailable. Now there is a new story of hope and promise to tell about Africa—a
story of a future where children who need the vaccine most will have a
chance at a healthy and happy life, free from the threat of severe rotavirus disease.
Today the GAVI Alliance approved rotavirus vaccine funding for 16 new
countries, 12 in Africa, including Angola, Burundi, Cameroon, Congo
DR, Djibouti, Ethiopia, Ghana, Madagascar, Malawi, Niger, Rwanda, and
Tanzania—and four other countries, including Armenia, Georgia, Moldova,
and Yemen. On July 17, 2011, Sudan became the
first African country to introduce rotavirus vaccines with GAVI
Alliance funding—just two years after the
World Health Organization recommended all countries introduce the
vaccine into their national immunization programs. People have heard
about Sudan’s introduction and have seen the news
around the world that vaccines against rotavirus are saving lives in
countries where children have access to them. They eagerly await the vaccine's arrival. |
|
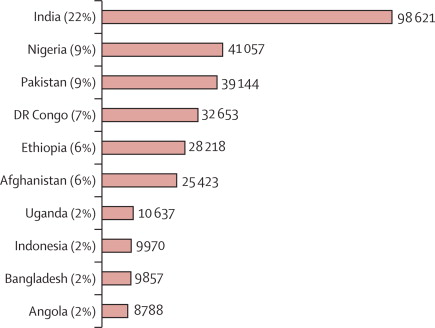
2008 estimate of worldwide rotavirus-associated mortality in children younger than 5 years before the introduction of universal rotavirus
vaccination programmes: a systematic review and meta-analysis
25 October 2011 - The Lancet Infectious Diseases, Early Online Publication
doi:10.1016/S1473-3099(11)70253-5 -
download pdf pdf
image: Countries with the greatest number of rotavirus-related deaths Rotavirus remains a major killer of children under five years of age worldwide, taking the lives of 453,000 children in 2008 according to the
latest estimates, published today in the Lancet Infectious Diseases journal. This translates into the staggering fact that more than 1,200 young children
will die from rotavirus diarrhea each day. Rotavirus-related deaths accounted for 37% of all diarrheal deaths and 5% of all deaths in children
under five years of age. One of every 260 children born each year will die from rotavirus diarrhea by their fifth birthday.
Tragically, approximately 95% of rotavirus deaths occurred in countries that are eligible to receive GAVI-support to introduce rotavirus vaccines. Five
countries–India, Nigeria, Pakistan, Democratic Republic of Congo, and Ethiopia–all GAVI-eligible, accounted for more than half of all rotavirus deaths globally. Introduction of effective and available rotavirus vaccines could substantially affect worldwide deaths attributable to diarrhoea. Our new
estimates can be used to advocate for rotavirus vaccine introduction and to monitor the effect of vaccination on mortality once introduced.
|
|
Diarrhea vaccines
In the developing world, where treatment can be hard to access and safe water is scarce,
diarrhea can be deadly. Prevention of diarrheal disease through immunization is a relatively new
intervention, but is becoming an essential and lifesaving part of diarrhea control strategies.
Vaccines against bacterial causes of diarrhea such as Shigella and enterotoxigenic
Escherichia coli (ETEC) are under development, and new vaccines against rotavirus
are increasingly available in low-resource settings. PATH is working with partners to increase access to existing rotavirus vaccines, develop new
rotavirus vaccines, and accelerate the development of other new vaccines against causes
of diarrhea. To learn more, please visit the PATH website. UNICEF is the world’s leading agency for vaccine procurement and procures vaccines and
immunization supplies on behalf of the GAVI
Alliance, which provides subsidized funding for rotavirus vaccine introduction in eligible countries.
Key resources
Below are some key documents on diarrhea vaccines. Please also visit our partners’ websites for more resources.
Other helpful websites
References
1 Parashar U, Hummelman EG, Bresee JS, Miller MA, Glass RI. Global illness and
deaths caused by rotavirus disease in children. Emerging Infectious Diseases.
2003;9(5):565-572. Photo: PATH/Mike Wang.
Reproduced from the PATH Resources for Diarrheal Disease Control website at
www.eddcontrol.org, [6 November, 2009].
A complete and up-to-date list of the following and related resources can be found at
Enhanced Diarrheal Disease Control Resource Center
Featured Resources
The effect of rotavirus vaccine on diarrhoea mortality
Melinda K Munos, Christa L Fischer Walker and Robert E Black
Approximately 39% of the global diarrhoea deaths in children aged 5 years
may be attributable to rotavirus infection. Two rotavirus vaccines were
recently introduced to the market, with evidence of efficacy in the USA,
Europe and Latin America. We sought to estimate the effectiveness of these
vaccines against rotavirus morbidity and mortality.
Rotavirus Vaccine — A Powerful Tool to Combat Deaths from Diarrhea
Mathuram Santosham, M.D., M.P.H.
Rotavirus infection, the leading cause of severe childhood diarrhea in both
developed and developing countries, results in over half a million deaths
each year.1 Currently, two rotavirus vaccines (Rotarix [GlaxoSmithKline
Biologicals] and RotaTeq [Merck]) are licensed in many countries and used
routinely in several. Until recently, available efficacy data were from
developed and developing countries with relatively low mortality rates among
children younger than 5 years of age.
The New England Journal of Medicine - Volume 362:358-360 - January 28, 2010 - Number 4
 pdf English 94 kb pdf English 94 kb
Anticipating new vaccines in the Americas (2004)
This editorial supports accelerated rotavirus vaccine introduction and
addresses related issues, such as health inequities, program sustainability,
and vaccine affordability.
Andrus, et al. Pan American Journal of Public Health.
16(6):369-370.
 pdf English 43
kb pdf English 43
kb
Cost-effectiveness of rotavirus vaccines (2005)
This paper reviews economic evaluations of Rotarix® and RotaTeq® and
offers suggestions for future analyses of cost-effectiveness.
Walker D, Rheingans R. Expert Review Pharmacoeconomics Outcomes
Research. 5(5):593-601.
 pdf English 309
kb pdf English 309
kb
Key facts about rotavirus disease and vaccines (2006)
Basic, essential information about rotavirus disease and vaccines against
rotavirus.
PATH
 pdf English 24
kb | pdf English 24
kb |
 pdf Español 32
kb | pdf Español 32
kb |
 pdf Russian 174
kb pdf Russian 174
kb
The promise of new rotavirus vaccines (2006)
This editorial summarizes findings from successful safety and efficacy studies
of Rotarix® and RotaTeq® vaccines, while also emphasizing the need for
clinical trials to evaluate the vaccines’ performance in developing country
settings.
Glass R, Parashar U. New England Journal of Medicine.
354(1): 75-77.
 pdf
English 694 kb | pdf
English 694 kb |
 pdf Español
230 kb | pdf Español
230 kb |
 pdf
Français 232 kb | pdf
Français 232 kb |
 pdf Português
247 kb | pdf Português
247 kb |
 pdf
Russian 245 kb pdf
Russian 245 kb
Rotavirus and severe childhood diarrhea (2006)
Study authors reviewed literature on hospitalizations related to severe
diarrhea and rotavirus and found that, while diarrheal disease incidence has
reduced in recent years, due in part to improved hygiene practices, incidence
of rotavirus infection has continued to increase.
Parashar U, Gibson C, Bresee J, Glass R. Emerging Infectious Diseases.
12(2):13-17.
 pdf English
141 kb pdf English
141 kb
Rotavirus: Questions and answers (2006)
Expanded information on rotavirus incidence and interventions, including
vaccines.
PATH
 pdf English 34
kb | pdf English 34
kb |
 pdf Español
41 kb pdf Español
41 kb
Rotavirus Vaccine Program
This collaboration between PATH, the World Health Organization (WHO), and the
US Centers for Disease Control and Prevention (CDC) aims to accelerate the
availability of rotavirus vaccines in the developing world.
Safety and efficacy of an attenuated vaccine against severe
rotavirus gastroenteritis (2006)
This study evaluated the safety and efficacy of GSK’s rotavirus vaccine (Rotarix®),
with a focus on determining risk of intussusception.
Ruiz-Palacios G, Pérez-Schael I, Velázquez F, et al. New England Journal
of Medicine. 354(1):11-22.
 pdf
English 233 kb | pdf
English 233 kb |
 pdf Español
233 kb | pdf Español
233 kb |
 pdf Français
244 kb | pdf Français
244 kb |
 pdf
Russian 253 kb pdf
Russian 253 kb
Safety and efficacy of a pentavalent human–bovine (WC3) reassortant
rotavirus vaccine (2006)
This study evaluated the safety and efficacy of Merck’s rotavirus vaccine (RotaTeq®),
with a focus on determining risk of intussusception.
Vesikari T, Matson D, Dennehy P, et al. New England Journal of Medicine.
354(1):23-33.
 pdf
English 1.17 mb | pdf
English 1.17 mb |
 pdf Español
216 kb | pdf Español
216 kb |
 pdf
Français 221 kb | pdf
Français 221 kb |
 pdf Russian
254 kb pdf Russian
254 kb
General information
Overview of RotaTeq® human-bovine reassortant rotavirus vaccine
(2005)
This presentation reported on studies of the safety and efficacy of the
RotaTeq® vaccine manufactured by Merck.
Shaw A, Heaton P. Merck & Co., Inc.
 pdf English 182
kb pdf English 182
kb
Proceedings of the Sixth International Rotavirus Symposium (2005)
An overview of the symposium’s sessions, presentations, and discussions.
The Albert B. Sabin Vaccine Institute
 pdf English 799
kb | pdf English 799
kb |
 pdf Español 820
kb pdf Español 820
kb
Rotarix® (2005)
This presentation from the director of worldwide medical affairs at GSK offers
a profile of Rotarix® and outlines its potential value.
De Vos B. Presented at: GlaxoSmithKline (GSK) R&D Day, London.
 pdf English 2.2
mb pdf English 2.2
mb
RotaShield® vaccine and intussusception Q&A
Answers to common questions about RotaShield®, an earlier vaccine against
rotavirus, and the decision of the CDC Advisory Committee on Immunization
Practices to no longer recommend it for use.
CDC
 pdf English 59
kb pdf English 59
kb
Rotavirus fact sheet (2005)
General information about rotavirus disease.
CDC
 pdf English 19
kb | pdf English 19
kb |
 pdf Español 23
kb pdf Español 23
kb
Treatment guidelines
Acute intussusception in infants and children. Incidence, clinical
presentation and management: A global perspective (2002)
This report from the WHO estimates global incidence of acute intussusception
in developing countries, the condition’s clinical presentation, and trends in
clinical management.
World Health Organization (WHO) Department of Immunization, Vaccines,
and Biologicals
 pdf English 317
kb pdf English 317
kb
Draft recommendations for pentavalent bovine-human rotavirus
vaccine (2006)
This presentation outlines recommendations of the CDC's Advisory Committee on
Immunization Practices for introduction of Merck's rotavirus vaccine, RotaTeq®,
into the routine US immunization schedule.
CDC Advisory Committee on Immunization Practices Rotavirus Working Group
English PowerPoint presentation 174 kb
Generic protocols for (i) hospital-based surveillance to estimate
the burden of rotavirus gastroenteritis in children and (ii) a community-based
survey on utilization of health care services for gastroenteritis in children
(2002)
WHO developed this guide for use in country activities aimed at collecting
data on local rotavirus disease burden.
WHO Department of Immunization, Vaccines, and Biologicals
 pdf English 1.5
mb | pdf English 1.5
mb |
 pdf Español 1.5
mb pdf Español 1.5
mb
Rotarix® international data sheet (2004)
This informational sheet contains prescription information for administration
of Rotarix® in Latin American countries, among others.
GSK
 pdf English
67 kb pdf English
67 kb
RotaTeq® package insert (2006)
This document provides information on and directions for administration of
Merck’s rotavirus vaccine.
Merck & Co., Inc.  pdf English 132
kb pdf English 132
kbResearch
Global Illness and Deaths Caused by Rotavirus Disease in Children
Parashar UD, Hummelman EG, Bresee JS, Miller MA, Glass RI. Emerg Infect Dis
[serial online] 2003 May.
Epidemiology of rotavirus diarrhoea in Africa: A review to assess
the need for rotavirus immunization (1998)
This study reviewed the epidemiology and disease burden of rotavirus diarrhea
among children at hospitals and clinics in African countries. The long-term
review was conducted from 1975 to 1992.
Cunliffe NA, Kilgore PE, Bresee JS, et al. Bulletin of the World
Health Organization. 76(5):525-537.
 pdf English 562
kb pdf English 562
kb
The epidemiology of rotavirus diarrhea in Latin America:
Anticipating new vaccines (2004)
This paper outlines a literature review performed to assess the disease burden
and epidemiology of rotavirus diarrhea in Latin America.
Kane E, Turcios R, Arvay M, et al. Pan American Journal of Public
Health. 16(6):371-377.
 pdf English 100
kb pdf English 100
kb
Evaluation of anatomic changes in young children with natural
rotavirus infection: is intussusception biologically plausible? (2004)
This study explores the plausibility of intussusception caused by natural
rotavirus infection.
Robinson C, Hernanz-Schulman M, Zhu Y, et al. Journal of Infectious
Diseases. 189:1382-1387.
 pdf English 300
kb pdf English 300
kb
Global illness and deaths caused by rotavirus disease in children
(2003)
This seminal paper estimates global incidence of rotavirus disease and related
deaths, based on a review of studies published from 1986 – 2000.
Parashar U, Hummelman E, Bresee J, et al. Emerging Infectious
Diseases. 9(5).
 pdf English 742
kb pdf English 742
kb
Hospitalizations associated with rotavirus diarrhea in the United
States, 1993 through 1995: Surveillance based on the new ICD-9-CM
rotavirus-specific diagnostic code (1998)
This study examined trends in rotavirus-associated hospitalizations among US
children.
Parashar U, Holman R, Clarke M, et al. Journal of Infectious
Diseases. 177:13-17.
 pdf English 153
kb pdf English 153
kb
Review of data from the REST and other Phase III studies of the
pentavalent human-bovine reassortant rotavirus vaccine, RotaTeq® (2006)
This presentation from the senior director of clinical research at Merck
Research Laboratories presents data on safety and efficacy clinical trials of
Merck’s rotavirus vaccine. Presented at the CDC Advisory Committee on
Immunization Practices meeting; Atlanta, GA.
Heaton P
English PowerPoint presentation
updated: 24 April, 2014
|